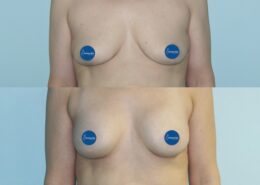
Breast implants have been used in aesthetic and reconstructive surgery for over 60 years. The first implants were introduced in the early 1960s, and since then, implant technology, materials, and safety standards have evolved significantly.
Modern breast implants differ remarkably from earlier models. While early implants often had very smooth surfaces and their contents and shells did not meet today’s safety standards, many changes have been made to manufacturing over the decades. Various fillers and surface treatments were used in past years, many of which have since been removed from production.
Why were textured implants introduced?
In the 1980s, textured (rough-surfaced) implants began to be widely used. At the time, it was believed that a textured surface would help the implant adhere better to the tissues and might reduce the risk of capsular contracture (the formation of excessive scar tissue).
However, it was later discovered that certain types of heavily textured implants could excessively irritate the patient’s tissues from the inside. The body may react by forming a thicker, multi-layered capsule around the implant. Serous fluid can accumulate between these capsule layers, and in rare cases, textured implants have been linked to BIA-ALCL—Breast Implant-Associated Anaplastic Large Cell Lymphoma.
BIA-ALCL is not breast cancer; it is a rare tumor of the immune system that usually develops in the capsule tissue surrounding the implant. According to US FDA data, BIA-ALCL occurs more frequently in patients with textured implants than in those with smooth-surfaced implants.
How rare is BIA-ALCL?
BIA-ALCL is very rare. Risk assessments vary by country and depend on the type of implant and the manufacturer. Based on published data, the lifetime risk for patients with textured implants is estimated to be approximately 1:2,207 to 1:86,029. In percentages, this is about 0.001–0.045%.
It is important to understand that the risk is not the same for all implants. Most attention has focused on certain heavily textured, or macro-textured, implants. Some of these have been removed from the market or recalled. For example, the FDA previously requested the recall of Allergan BIOCELL textured implants due to the risk of BIA-ALCL.
The implant might be intact, but the tissue around it might not be
One of the main issues with old textured implants is that patients may not have received sufficient information regarding the potential need for replacement.
A mammogram or ultrasound may show that the implant itself is intact. However, this does not always mean that the surrounding tissues and capsule are completely problem-free. A heavily textured surface can cause chronic tissue irritation, inflammatory reactions, or the development of a thicker capsule over the years.
Therefore, the evaluation should not only focus on whether the implant has ruptured. It is essential to assess the type of implant, its age, the surface type, the patient’s symptoms, and the condition of the tissues surrounding the implant.
Modern nanotechnology surface implants
In recent years, there has been an increasing preference for implants with a light structure or a nanotechnology surface. The aim of such implants is to reduce excessive tissue irritation and provide a safer interaction with the body.
One example is Motiva implants, of which over five million have been placed worldwide, with no confirmed cases of BIA-ALCL linked to primary Motiva implant placements to date.
Experience at Christinas Clinic
At Christinas Clinic, we have used only nanotechnology, smooth, or lightly structured surface implants for the past 21 years. These do not require automatic replacement 10 years after primary placement. The patient’s health and their body’s reaction to the foreign body (the implant) must always be evaluated.
If you have breast implants, we recommend checking your implant passport for the manufacturer, model, year of placement, and surface type. If the implant is textured and was placed more than 10 years ago, it is worth coming for a consultation to evaluate the condition of the implants and the surrounding tissues.
It is important to know that while a mammogram or ultrasound may show an intact implant, it does not guarantee that the capsule and tissues are problem-free. With old textured implants, issues can arise specifically in the surrounding tissues.
Implant replacement should be considered especially if the implants are old textured models or if symptoms such as pain, swelling, fluid accumulation, change in shape, asymmetry, or thickening of the capsule have occurred.
If you have old textured breast implants, check your implant passport and come for a consultation if necessary. At Christinas Clinic, we help evaluate the condition of your implants and offer modern solutions for their replacement.
Replacement of implants placed at Christinas Clinic: from €3,990
Replacement of implants placed at another clinic: from €4,590
 https://www.christinasclinic.ee/wp-content/uploads/motiva-qinsidesafety-130-BS-1.jpg
1080
1920
cc
https://www.christinasclinic.ee/wp-content/uploads/Christinas-Clinic-LOGO.png
cc2026-02-05 09:24:492026-02-05 09:24:49If a Woman Is Not Happy with Her Breasts
https://www.christinasclinic.ee/wp-content/uploads/motiva-qinsidesafety-130-BS-1.jpg
1080
1920
cc
https://www.christinasclinic.ee/wp-content/uploads/Christinas-Clinic-LOGO.png
cc2026-02-05 09:24:492026-02-05 09:24:49If a Woman Is Not Happy with Her Breasts https://www.christinasclinic.ee/wp-content/uploads/motiva-qinsidesafety-130-BS-1.jpg
1080
1920
cc
https://www.christinasclinic.ee/wp-content/uploads/Christinas-Clinic-LOGO.png
cc2026-02-05 09:24:492026-02-05 09:24:49If a Woman Is Not Happy with Her Breasts
https://www.christinasclinic.ee/wp-content/uploads/motiva-qinsidesafety-130-BS-1.jpg
1080
1920
cc
https://www.christinasclinic.ee/wp-content/uploads/Christinas-Clinic-LOGO.png
cc2026-02-05 09:24:492026-02-05 09:24:49If a Woman Is Not Happy with Her Breasts