https://www.christinasclinic.ee/wp-content/uploads/mommy-e1579258253917.jpg
818
1200
cc
https://www.christinasclinic.ee/wp-content/uploads/Christinas-Clinic-LOGO.png
cc2020-02-11 11:42:022026-02-20 14:18:12Mommy makeover – a service actually needed or a successful advertising campaign?
https://www.christinasclinic.ee/wp-content/uploads/mommy-e1579258253917.jpg
818
1200
cc
https://www.christinasclinic.ee/wp-content/uploads/Christinas-Clinic-LOGO.png
cc2020-02-11 11:42:022026-02-20 14:18:12Mommy makeover – a service actually needed or a successful advertising campaign?Motiva implants mark the new era
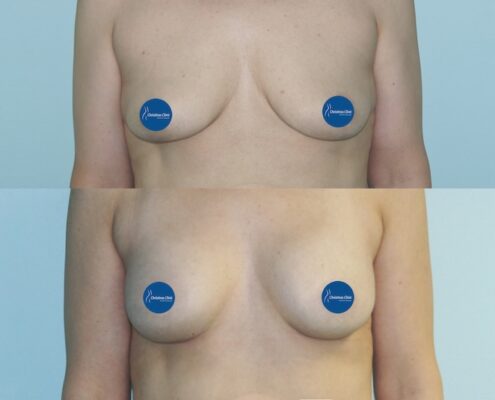
Christinas Clinic has widened the choice of implants with innovative Motiva products.
We often hear about new or improved implants entering the market, yet during the past 20 years no fundamental changes in production have taken place. Motiva’s progress in finding innovative solutions, therefore, deserve special attention. Major changes, which will be overviewed in this segment, have been made on the level of the implant’s shell, form and safety.
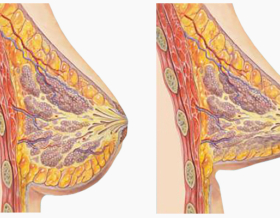
The overall quality of the implant largely depends on the implant shell. The shell consists of several layers, which have different features and functions. The layer that prevents silicone leakage is usually imperceptible, and therefore the possible defects are not detectable. Motiva has given colour to the layer, which makes it easy for the surgeon to identify if the implant is safe for the patient.
Another significant feature is the roughness of the surface layer. So far, salt or sugar has been used to create the needed roughness. Manchester University’s scientist Dr. Bayat, in collaboration with Motiva has developed a new surface designed to improve their biocompatibility, using nanotechnology. The new technology reduces the risk of capsular contracture.
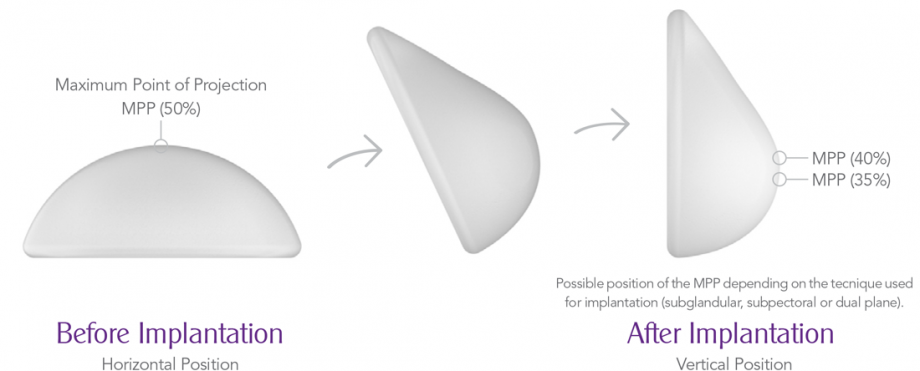
Another innovation concerns the shape of the implants. Anatomical implants may start rotating in the body after implantation. Motiva Ergonomix are made to adapt the shape after implantation. While the implants are round, the maximum point of projection shifts to the lower pole when the patient stands up. Similar to the natural breast, the projection point moves to the center of the breast when the patient lies down.
To ensure product safety, Motiva has introduced Q Inside Safety Technology. At a request, the implant comes with a micro-transponder programmed with a serial number that can be accessed by a handheld reader when waved over the breast area. The number corresponds with a secure, online database, which stores information about the product, including the warranties.
The implants come with an up to ten-year product warranty for all complications, including capsule contraction (Baker scale 3 and 4). For the first time, beginning with this cycle, the warranty is issued by a third party, the insurance company. It means that the patient stays insured even if the production company disappears from the market (e.g. PIP case).
The materials used by Motiva are all approved by both the European and the American (FDA) agencies. A serious indication of high standards and quality is that in 2015 the FDA is monitoring the factory and production processes.
Book a consultation and ask more about Motiva implants!
Breast augmentation
Duration: 1 hour